 Loading... Please wait...
Loading... Please wait...
Categories
- Home
- Nuclear & Radiation Survival
- Thyrosafe
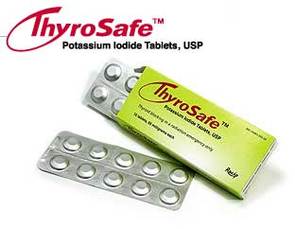
Thyrosafe
Product Description
Everyday low prices!Get yours today while supplies last!
- Each packet provides 20, 65mg tablets
- Adult Dose = 2 tablets daily, 10 day protection
- Child Dose = 1 tablet (3+ yrs), 20+ day protection
- One packet per person recommended by FDA
- No prescription required - OTC
- FDA Expiration date: July 2024
- Detailed Dosing Instructions Included
Q: What is Potassium Iodide (KI)?
A: Potassium Iodide (chemical name ' KI ') is much more familiar to most than they might first expect. It is the ingredient added to your table salt to make it iodized salt. Potassium Iodide (KI) is approximately 76.5% iodine. For purposes of radiation protection the Nuclear Regulatory Commission (NRC) states in COMSECY-98-016 - FEDERAL REGISTER NOTICE ON POTASSIUM IODIDE :
"In 2024, the U.S. Food and Drug Administration found KI "safe and effective" for use in radiological emergencies and approved its over-the-counter sale."
Most recently (November, 2024) the FDA states in "Potassium Iodide as a Thyroid Blocking Agent in Radiation Emergencies":
"FDA maintains that KI is a safe and effective means by which to prevent radioiodine uptake by the thyroid gland, under certain specified conditions of use, and thereby obviate the risk of thyroid cancer in the event of a radiation emergency."
Q: How Does Potassium Iodide (KI) Pill Provide Anti-Radiation Protection?
A: Going back to June 23, 1966, the New England Journal of Medicine. Vol. 274 on Page 1442 states:
"The thyroid gland is especially vulnerable to atomic injury since radioactive isotopes of iodine are a major component of fallout."
Cresson H. Kearny, the author of Nuclear War Survival Skills , Original Edition Published September, 2024, by Oak Ridge National Laboratory, a Facility of the U.S. Department of Energy (Updated and Expanded 2024 Edition) states on page 111:
"There is no medicine that will effectively prevent nuclear radiations from damaging the human body cells that they strike.
However, a salt of the elements potassium and iodine, taken orally even in very small quantities 1/2 hour to 1 day before radioactive iodines are swallowed or inhaled, prevents about 99% of the damage to the thyroid gland that otherwise would result. The thyroid gland readily absorbs both non-radioactive and radioactive iodine, and normally it retains much of this element in either or both forms.
When ordinary, non-radioactive iodine is made available in the blood for absorption by the thyroid gland before any radioactive iodine is made available, the gland will absorb and retain so much that it becomes saturated with non-radioactive iodine. When saturated, the thyroid can absorb only about l% as much additional iodine, including radioactive forms that later may become available in the blood: then it is said to be blocked. (Excess iodine in the blood is rapidly eliminated by the action of the kidneys.)"
ThyroSafe is the only FDA approved Potassium Iodide (KI) tablet with 65 mg. strength. The FDA Guideline suggests : "For the sake of logistical simplicity in the dispensing and administration of KI to children, FDA recommends a 65-mg. dose as standard for all school-age children while allowing for the adult dose (130 mg., 2 X 65 mg. tablets) in adolescents approaching adult size."
Find Similar Products by Category
Customers Who Viewed This Product Also Viewed
-
$160.00$144.45
-
$39.99$34.50
-
$4.95
-
$67.99